
Materialise Receives First-Ever 510(k) Clearance for Anatomical Model 3D Printing Software
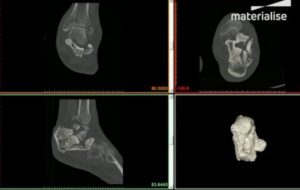
3D printing specialist Materialise recently received FDA 510(k) clearance for its Mimics inPrint software. Mimics inPrint is said to use to create 3D anatomical models from medical image data and output those models to a 3D printer at the point-of-care. The physical models created with Mimics inPrint assist practitioners with patient diagnostics, consultation, and planning complex procedures. According to a company press release, Mimics inPrint is already used by 16 of the top 20 hospitals to provide personalized patient care at the point-of-care.
In the press release, Materialise CEO Wilfried Vancraen stated:
Materialise has nearly three decades of experience in developing certified medical solutions that create a better and healthier world. The FDA clearance for our Mimics inPrint software will support the adoption of 3D planning and printing in U.S. hospitals and the creation of point-of-care 3D printing facilities.
According to Frank J. Rybicki, MD, PhD and Chief of Medical Imaging at Ottawa Hospital:
This milestone for Materialise serves as a benchmark for the clinical implementation of 3D printing for physicians creating 3D models at the point-of-care.
Materialise describes itself as having 27 years of 3D printing experience and according to USPTO public database records is currently the assignee of 34 issued patents and 106 patent applications dating from 2004 to the present.