The Federal Circuit has affirmed that Eagle Pharmaceuticals, Inc. did not infringe Par Pharmaceutical, Inc. patents, easing Eagle’s path to market a generic competitor to Par’s Vasostrict®
product. Par had alleged that Eagle’s abbreviated new drug application (ANDA) infringed U.S. Patent Nos. 9,744,209 and 9,750,785, both titled “Vasopressin formulations for use in treatment of hypotension.” An example molecular structure is shown here:
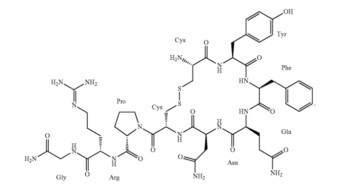
Some of Par’s arguments had alleged that because the pH of Eagle’s products would be so close to Par’s claimed ranges for pH, inevitable drift in pH would occur over time, causing infringement. In affirming, Chief Judge Moore explained that the District Court had not committed clear error in finding otherwise. The infringement inquiry “begins and ends” with Eagle’s ANDA specification, so predicting future drift cannot show infringement.
Thus, the Federal Circuit upheld the District Court’s decision against Par Pharmaceutical and affirmed the finding of no infringement. The Federal Circuit’s decision is available here.
Rory R. Lootsma (New York Bar Admission Pending)
