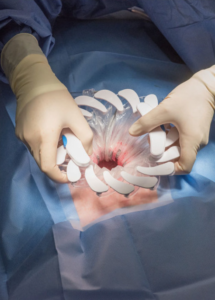
FDA Clears New Surgery Infection Control System
The FDA recently cleared Prescient Surgical’s CleanCisionTM Wound Retraction and Protection System (CleanCisionTM), a retractable device designed to prevent surgical site infection. According to Prescient Surgical, CleanCisionTM is “a novel, first-in-class, advanced intraoperative infection control system” that utilizes an active cleansing technology.
Traditionally, practices for preventing surgical site infections (SSIs) involve administering drugs, controlling patient’s blood glucose level, maintaining optimal temperature and tissue oxygenation, using skin prep agents, and using plastic wound protectors.
The press release states that unlike traditional methods, ClearCision combines wound protection and wound cleansing. Retractable plastic sleeves protect the wound while providing direct access to the surgical site. Irrigation system continuously cleans the wound edge using sterile irrigant solution and removes contaminant using suction. Insoo Suh, cofounder of Prescient Surgical, noted that ClearCision is “a proactive approach to clearing contamination during surgery.”
The press release also notes that SSIs can be caused by bacteria entering the surgical incision site, and lead to significant financial burdens on teh healthcare system.
Jonathan Coe, cofounder, president, and CEO of Prescient Surgical, said in the press release that the company is “initially focusing on abdominal surgery and particularly colorectal surgery, where the risk, frequency and severity of [SSI] is high and the need is acute.”